Introduction
Viscum album L. (white mistletoe or European mistletoe) is a semi-parasitic plant that is common in South-West Asia and most European countries, including Ukraine. It inhabits numerous decorative and economically important tree species. V. album like many other mistletoes is believed to cause growth retardation, premature defoliation, reduction of photosynthetic potential and subtle changes in the water-carbon balance of host trees, which inevitably leads to a decrease in the resistance of woody plants to other damaging factors (Barbu 2012, Sanguesa-Barreda et al. 2013, Alvarado-Rosales and Saavedra-Romero 2021, Thomas et al. 2022). This can be a serious threat to host plants.
There are various methods of controlling mistletoe (pruning of infected branches, removal of infected trees, use of systemic herbicides, etc.), but all of them significantly affect the host plants (Varga et al. 2012a, Watson 2019, Mudgal et al. 2022). There are some promising methods of biological control, but they require further elaboration and development and/or the use of special equipment (Krasylenko et al. 2023). The methods that target pollinators and dispersers are unacceptable because they lead to significant negative environmental consequences. Hyper parasitism (parasitism of one mistletoe on another) causes overloading of the host plant and can lead to its death. However, the use of natural antagonists, such as bacteria and fungi, is considered a more promising option (Karadžić et al. 2004, Kotan et al. 2013, Chen et al. 2018).
There are several genera of pathogenic fungi associated with mistletoe diseases, among which Botryosphaeria visci is relatively more aggressive as it causes mistletoe dieback (Kotan et al. 2013, Karadžić et al. 2004, Kahle-Zuber 2008). Traditional research on B. visci to date has focused on taxonomic profiling, particularly based on molecular, macro- and microscopic studies (Phillips et al. 2008, Poczai et al. 2015, Varga et al. 2014). It should be noted that studies of the growth of this pathogenic fungus in vitro can reveal some important physiological features. However, research on B. visci is fragmentary and almost none has been conducted on the factors influencing its in vitro growth. Studies of B. visci and the disease it causes have included culturing the fungus on various media for growth and sporulation (Varga et al. 2012b, Varga et al. 2013) and examining temperature parameters (Zlatković et al. 2016, Tkaczyk and Sikora 2020). Due to the high pathogenicity of this fungus, several studies have focused on evaluating it as a promising candidate for the biological control of mistletoe (Karadžić et al. 2004, Varga et al. 2012a, Kotan et al. 2013, Tkaczyk and Sikora 2020). At the same time, it must be remembered that mistletoe is part of ecosystems, and its complete removal reduces biodiversity. Thus, the greatest potential for the use of B. visci as a biocontrol agent for Viscum album should be expected in the protection of commercial plantations and orchards.
The purpose of this work was to isolate and identify the fungus Botryosphaeria visci associated with Viscum аlbum L . from Sorbus aucuparia, to elaborate its introduction and growth in vitro, as well as to test the pathogenicity of the isolated fungus.
Sampling
Diseased parts of common mistletoe (stems and leaves) were collected in Ukraine (Sumy region) in August 2021, where a mass appearance of white mistletoe was observed. Samples of mistletoe plants were taken from host-tree Sorbus aucuparia L., usually moderately damaged due to infection with the above-mentioned plant pathogen (from 10 to 18 bushes). From these trees, ten shoots were taken with visible black pycnidia and preserved as dry herbarium samples (stored in paper bags, with the sampling information).
Identification
The fungus was removed with a sterile needle and transferred to a small drop of twice-distilled water on a clean slide and covered with a cover glass. Identification of samples was based on morphological characteristics of conidia from infected host materials under an Axiostar microscope, (Zeiss, Germany), photographs were taken using the AxioCam MRc5 digital camera (Zeiss, Germany) and the AxioVision 4.7 software (Zeiss, Germany) and microscope Nicon SMZ18 SHR Plan Apo with camera Nikon DS-FI3 and software NIS-Elements D5.03.02 (Build 1545). A total of 20 conidiomata and 50 conidia were measured to calculate the mean size and standard deviation (SD). The taxonomic affiliation of the fungus was determined by a combination of morphological and cultural features (Phillips et al. 2013).
Isolation
Isolation was carried out by transferring conidia, after surface sterilization with 3% hydrogen peroxide for 1-2 minutes on potato-dextrose agar (PDA) (Varga et al. 2012a). Single germinating spores were transferred to fresh PDA plates and incubated at 25 °C in the dark. Maternal cultures of the introduced fungi strains were kept on PDA slants at 4 ºC before use. A total of 8 fungal isolates were obtained in pure culture. However, 5 isolates were identified as Botryosphaeria visci. These pure fungal cultures were deposited in the collection of microorganism strains and plant lines at the Institute of Food Biotechnology and Genomics at the National Academy of Sciences of Ukraine. The B. visci strain IFBG 96 with the best growth characteristics was selected for further research in this study.
Cultivated media
The choice of media for cultivation was based on the information available in the literature about the growth of the fungus; for a more effective comparison and reproduction of the results, the synthetic media that are most often used were chosen (Varga et al. 2012b, 2013, 2014).
Morphological characteristics of vegetative mycelia and evaluation of the isolate’s growth were provided using the tested media: PDA, Difco, USA; Sabouraud dextrose agar (SDA), Difco, USA; glucose-peptone-yeast agar (GPYA), containing, in g L -1: 25.0 glucose, 3.0 yeast extract, 2.0 peptone, 1.0 K 2HPO 4, 1.0 KH 2PO 4, 0.25 MgSO 4·7H 2O, and 15.0 agar (pH 6.0); oatmeal agar (OMA), Difco, USA.
The liquid media used (PDB, GPYB, SDB, OMB) had the same composition as above except for agar. Media pH was adjusted before autoclaving. The media were distributed to 9 cm sterile Petri dishes. Flat plates were inoculated with a mycelial block of the fungus cut from GPYA Petri dishes using a sterile 8 mm diameter drill bit at the stage of active mycelial growth. The morphology of the cultures and pigment production were assessed visually and noted after 1 week of growth on the test media. Colony colors (surface and reverse) were rated according to Rayner’s color chart (1970).
For liquid suspension culture, 100 mL media was inoculated (in 250 mL vol. cap. flasks) with three mycelial wells cut from GPYA Petri dishes as above. Cultivation on solid and liquid media was carried out at 25 ºC in the dark at static conditions.
Biomass dry weight
On the 14 th day of growth, the mycelium was separated from the liquid medium by filtering through Whatman filter paper No. 4, washed with distilled water, and dried to constant weight at 105 °C. Fungi biomass was estimated by absolutely dry weight (a.d.w.).
Pathogenicity tests
Reinfections in laboratory conditions were performed with four fresh sterilized (in 3% H 2O 2, 2 minutes) healthy mistletoe leaves which were placed into Petri dishes with a colony of B. visci grown on solid media (OMA and SDA) and in a vial (culture mattress) with the mycelial matt of B. visci grown in SDB. The fungal isolate was grown on these suitable growth media at 25 °C under light during the day for spore formation and in the dark at night. Repeated infections in laboratory conditions were carried out in three replicates.
Tests for pathogenicity in field conditions were carried out by spraying with culture liquid. A spray bottle was used with a suspension collected after 21 days of growth of B. visci in SDB medium. A sterile wire loop was used to scrape the conidia and bring them to a suspension. The suspension was filtered through a muslin cloth, and the collected filtrate was serially diluted to 1 × 10 7. A haemocytometer was used to adjust the spore concentration. About 30 live mistletoe bushes were sprayed with the resulting liquids using manual sprayers. These bushes grew on different host trees. The distance between the treated V. album plants was between 2 and 15 m. All mistletoe bushes were located at a height below 2 m. Sterilized distilled water was used as a negative control. Additionally, some mistletoe leaves were mechanically damaged with a blade to allow liquid contact with parenchymal cells. The processing was carried out in autumn (September). The conditions were quite favourable for the growth of B. visci, since the species is able to grow actively until the onset of cold weather (Tkaczyk and Sikora 2020). The average daily temperature was +13.5 °C and the humidity was 63%. Over the following two months, the impact on mistletoe was recorded. According to the formation of pycnidia or of their absence on leaves, shoots or berries of mistletoe, pathogenicity was evaluated as positive or negative, respectively. Re-isolations were performed from infected leaves and shoots (Varga et al. 2014), and the resulting isolates were identified as previously described.
Statistical analysis
The experimental values were expressed as mean ± SD (standard deviation) of at least three replicates. Statistical significance analysis was performed by Fisher Least Significant Difference (LSD) test at 0.05 probability level using computer Software SPSS 24.0 (SPSS Inc, Chicago, IL, USA).
Results
The fungus was isolated from fresh collections of the European mistletoe, Viscum album from the host-tree Sorbus aucuparia. Based on the morphological characteristics of the conidia from the infected host material, it was established that this is an anamorph Sphaeropsis visci of the ascomycete Botryosphaeria visci (Fig. 1). Below is a taxonomic description of the fungus according to Ukrainian samples. Its morphological features are described in detail and illustrated with photomicrographs obtained using light microscopy. Taxonomic placement and author citations are provided according to Index Fungorum Database (2023).

Fig. 1.Botryosphaeria visci and infected diseased mistletoe: A – infected European mistletoe ( Viscum album L.) on Sorbus aucuparia L.; B-E – pycnidia forming on leaves and berries of mistletoe; F – conidia of the pathogenic fungus; G – conidiogenous cells and conidia of pycnidia isolated from a colony on Sabouraud dextrose agar medium; H – mycelium of B. visci. Scale bars: D = 1 mm, E = 0.1 mm, F = 10 µm, G = 200 µm, H = 100 µm.
Botryosphaeria visci (Kalchbr.) Arx & E. Müll. Beitr. Kryptfl. Schweiz 11(1): 41, 1954. Syn: Phaeobotryon visci (Kalchbr.) Höhn., Sber. Akad. Wiss. Wien, Math.- naturw. Kl., Abt. 1 128(7-8): 591. 1919.
Anamorph: Sphaeropsis visci (Alb. & Schwein.) Sacc., Michelia 2(6): 105, 1880.
Descriptions: Conidiomata pycnidia, initially submerged in leaf tissue, erupt and become superficial at maturity, scattered to gregarious, globose, dark brown to black, approximately 300 µm in diameter. Pycnidia unilocular single-nested, with dark layered walls, the inner wall is formed of dark brown angular cells, textura angularis. Pseudoparaphyses 4-5 µm wide, hyaline, cylindrical, aseptic, unbranched. Conidiogenous cells are located between pseudoparaphyses, hyaline or yellowish-brown, 8-11 × 5.5-6 µm, with periclinal thickenings. Pycnoconidia are brown, oval or ovoid, 28-42 × 17-22 µm, with truncate or obtuse base and obtuse apex, thick-walled with a fine warty surface texture inside.
Sexual morph: not observed.
Culture characteristics:B. visci isolate IFBG 96 was found to grow on all semi-solid media but with variations in mycelial growth rate and colony morphology. Colonies on PDA, GPYA, and SDA had appressed moderately dense matted mycelium, with irregular sparse zonal aerial mycelium or rosette-like appearance, wavy edge not reaching the edge of the Petri dish for 4 weeks (Fig. 2A-C). The surface of the colony on PDA was smoke-grey to black, reverse side was dark black (Fig. 2A). Colony surface on GPYA was pale mouse-grey, reverse side mouse-grey to dark mouse-grey (Fig. 2B). The surface of the colony on SDA was apricot to chestnut; reverse side umber to chestnut (Fig. 2C). Colonies on OMA were off-white with dense aerial mycelium to cottony, covering the dish after 7 days, reverse side black (Fig. 2D). Pycnidia were unevenly distributed on the surface of the SDA medium. The first pycnidia appeared beside the inoculation block, then they sporadically covered the media surface.
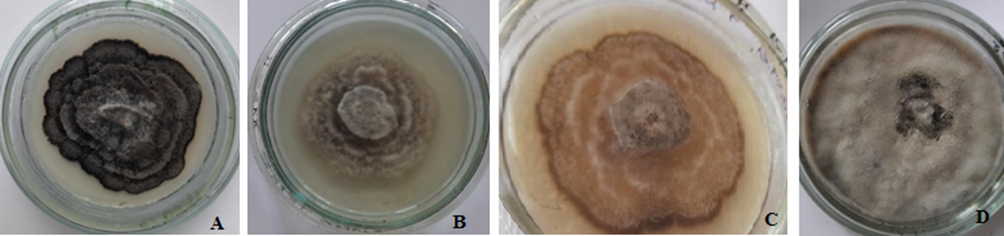
Fig. 2. Morphological differences of Botryosphaeria visci colonies grown on different media: A – potato dextrose agar (PDA), B – glucose-peptone-yeast agar (GPYA), C – Sabouraud dextrose agar (SDA), D – oatmeal agar (OMA).
Colony morphology of B. visci on liquid media was similar to that on agar media. The surface mycelia of B. visci started to grow on the third day and during 14 days of cultivation completely covered the surface of all media in the flasks. The mycelium was downy, uniform in texture and consistency, white to greyish-green in colour, with airy hyphae on the surface of the liquid media (Fig. 3A). Liquid media also had a significant effect on mycelial growth of B. visci isolate IFBG 96 (Fig. 3B). The results showed that OMB was the most suitable liquid culture medium for the growth of B. visci. Moderate mycelial growth was observed in SDB medium followed by GPYB medium. Limited growth of mycelium was established in PDB. The final pH of the culture media varied from 4.7 to 6.0.
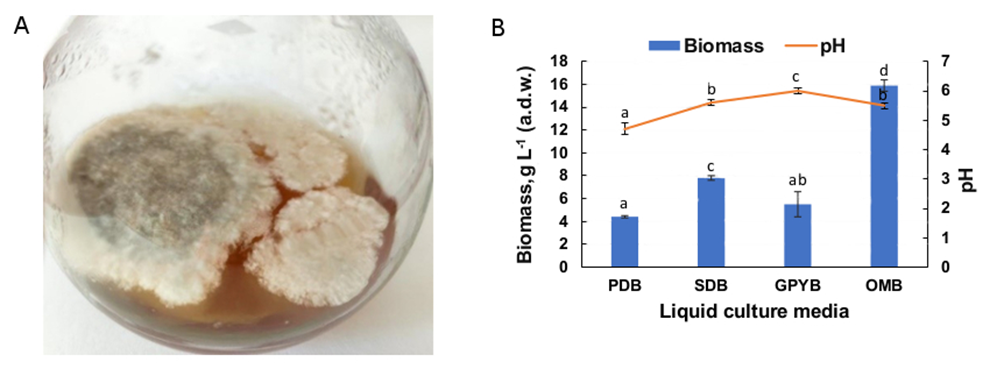
Fig. 3. The morphology of Botryosphaeria visci colony on the 10 th day of growth in GPYB medium (A), and biomass production of B. visci after 14 days of inoculation in liquid culture media (B): PDB − potato dextrose broth, SDB − Sabouraud dextrose broth, GPYB − glucose-peptone-yeast broth, OMB − oatmeal broth. Data are mean ± standard deviation, n = 3. Different letters indicate significant differences at P < 0.05 by Fisher`s LSD test.
Possibility of infection: Laboratory experiments of mistletoe pathogenicity were modelled on different nutrient media. A negative result of mistletoe leaf infection was obtained on solid agar media; no infection of mistletoe leaves placed on fungal colonies in Petri dishes on OMA (Fig. 4A) and SDA (Fig. 4B) media was detected. Neither did mechanical damage to V. album leaves before they were placed on fungal colonies in Petri dishes contribute to infection in our experiments. A positive result of infection of V. album leaves was obtained with B. visci grown in liquid medium. The formation of characteristic pycnidia on the mycelium of B. visci was visually observed starting from the 21st day of cultivation in Sabouraud dextrose broth. It should be noted that the highest concentration of pycnidia was found in the immediate vicinity of the leaves (Fig. 4C-D), followed by infection and development of infection on 45-50% of the leaf area of V. album.

Fig. 4. Possibility of mistletoe infection in the laboratory on 21 days on oatmeal agar (A), on Sabouraud dextrose agar (B), in Sabouraud dextrose broth (C, D), and in the field 60 days after treatment with B. visci liquid culture, obtained after growth in Sabouraud dextrose broth (E–I).
The experiment in the field showed that only a few plants had initial signs of infection after spraying with liquid culture of B. visci (Fig. 4E). Only previously damaged plants were infected. Their number was about 6.5% of all processed mistletoe bushes. Undamaged plants had no signs of infection.
The weakening of the host plant contributed to the development of the infection. After processing the samples, we noted mechanical damage to one of the host trees, which had a negative impact on the overall condition of the mistletoe. As a result, pycnidia formation was observed on 65% of inoculated leaves and 28% of inoculated shoots (Fig. 4F-H). At the same time, no damage was detected on the berries.
The results of the pathogenicity test in field conditions indicated that the isolated pathogen can infect V. album only at minimal effectiveness under the condition of mechanical pre-treatment and in already weakened mistletoe. Conidiomata isolated from the laboratory experiments under liquid conditions and from re-infested field conditions were re-isolated by implanting on PDA medium, thus fulfilling Koch’s postulates. All isolates were confirmed to belong to Botryosphaeria visci.
Discussion
The rapid spread of semi-parasitic mistletoe, which has become a serious pest of trees, requires the search for and development of simple, effective means of control (Mudgal et al. 2022). The literature supports the possibility of biotechnological interventions such as Sphaeropsis visci fungi as a biological control agent for mistletoe (Varga et al. 2012b, 2013, Tkaczyk and Sikora, 2020). These studies concern B. visci, obtained from Viscum album on host trees such as Acer saccharinum L. (Varga et al. 2012b, 2013), A. pseudoplatanus L. and Populus nigra L. (Varga et al. 2012a), as well as from pine plantations (Tkaczyk and Sikora 2020). To the best of our knowledge, this is the first report of an in vitro study of B. visci, isolated from V. album on the host tree Sorbus aucuparia L. which belongs to the category of secondary hosts (Krasylenko et al. 2020). The morphological characteristics of conidiomata (pycnidia) from collected samples of European mistletoe clearly identify the anamorph Sphaeropsis visci of ascomycete Botryosphaeria visci. Identification was based solely on morphological characteristics of conidia from infected host material, but molecular analysis of the fungus is also required. This fungus is an aggressive pathogen on mistletoe in various countries, for example in Romania (Sutton 1980), Croatia (Idžojtić et al. 2008), Ukraine (Phillips et al. 2008), Austria, the Czech Republic, Egypt (Phillips et al. 2013), Hungary (Varga et al. 2014, Poczai et al. 2015), Serbia, Luxemburg (Zlatković et al. 2016, Zlatković et al. 2015), China (Chen et al. 2018), and Poland (Tkaczyk and Sikora 2020).
Introduction of the fungus into the in vitro culture and investigation of its growth and physiology are necessary for a comprehensive study of the fungus. Despite the ecological and often physiological diversity, the specific adaptation to different woody hosts, species from the Botryosphaeriaceae family can be grown in culture conditions (Luque et al. 2005, Abdollahzadeh et al. 2009, Yang et al. 2017, Zhang et al. 2021, Wu et al. 2021). In this study, S. visci isolates belonging to Botryosphaeriaceae could be cultured on PDA, which is consistent with previous reports (Varga et al. 2012a, Varga et al. 2013, Varga et al. 2014). No ascomata were detected either on the host or in culture, which is consistent with previous studies (Varga et al. 2014, Chen et al. 2018, Tkaczyk and Sikora 2020).
Vegetative mycelial growth and colony morphology are important criteria for culture characterization. Growth and morphology of B. visci were strongly affected by the nutrient media probably due to the use of test media that differed in carbon sources. This result of our study confirms the observations of similar studies on the effect of the medium on fungus growth (Varga et al. 2012b, Varga et al. 2013). The isolated fungus grew on all solid and liquid media at different growth rates. To our knowledge, this is the first time that SDA and SDB media have been used for the isolation and cultivation of B. visci, and that the effect of different liquid media on B. visci growth has been evaluated by mycelial dry weight. Although PDA as well as PDB were commonly used for culturing the fungus in previous studies, this medium proved ineffective for our research because of the slow growth of B. visci. Fungal strains and isolates may differ in the degree of cultivation in different media compositions. At the same time, the use of natural environments can enhance the growth and/or sporulation of fungal pathogens. Rather, PDA was the first medium tested, but oatmeal medium is the most common and effective medium for B. visci cultivation. The OMA as well as OMB media in this study effectively supported the growth and sporulation of B. visci, which could be a low cost alternative for use in routine mycological and pathological studies involving B. visci isolates. Our results are consistent with previous studies that found good growth and sporulation of this fungus on OMA (Varga et al. 2012b, Varga et al. 2013). This finding indicates that B. visci prefers the oatmeal medium regardless of the fungal strain, probably due to the presence of a plant component in the medium.
Isolation and functional testing of pathogenic fungal effectors has an important role to play in developing new approaches to disease control, particularly the effective control of pathogen damage. The conducted phytopathogenic test of B. visci was positive in the case of liquid culture conditions and negative when tested on solid agar media. This can be explained by the fact that under the conditions of liquid cultivation, the isolate had a significantly larger mycelial surface and a larger volume of nutrient medium, which contributed to the intensive production of conidia and, accordingly, the formation of pycnidia. The formation of pycnidia by the fungus B. visci in the immediate vicinity of the host plant, followed by infestation, indicates a direct relationship with mistletoe. Some studies have also shown that the use of pathogenic fungi such as B. visci can be considered as a potential method for the biological control of European mistletoe pathogen (Varga et al. 2012a, Kotan et al. 2013, Tkaczyk and Sikora 2020). As a rule, fungal isolates in the form of fungal suspensions were tested for pathogenicity on the young leaves of white mistletoe by using polyethylene bags (Varga et al. 2012a, Tkaczyk and Sikora 2020) or by using a bottle sprayer in field conditions. According to our results, placing host samples on the surface of the fungal mycelium should be considered as an alternative, easy-to-perform test for phytopathogenicity only during product development phase that allows visual observation of the relationship between the pathogen and the host plant. The phytopathogenicity test of B. visci conducted in the field was positive only after mechanical pre-treatment, which is consistent with the findings of Karadžić et al. (2004). A low intensity of infection in our field conditions can be related both to deteriorating climatic conditions (a drop in temperature to negative values in autumn) and to a change in the physiological state of the white mistletoe, which, together with the host plant, suppresses its physiological activity for the winter (Agne et al., 2014, Mutlu et al., 2016). Abiotic factors (temperature, humidity, etc.) have also been identified as key factors for successful infection in studies by Varga et al. (2012a). However, the strongest effect after treating mistletoe plants with a mycopesticide was observed when the mistletoe was weakened or suppressed by previous exposure to chemicals (Baltazár 2016). Pre-treatment with herbicides is also proposed as a way to reduce the vital activity of mistletoe (Baltazár 2016), but this, in our opinion, contradicts the very idea of biocontrol. An alternative approach to suppress mistletoe growth by using bacterial or fungal isolates, such as Alternaria alternata and A. kiliense, as pathogenic for mistletoe, has also been proposed (Kotan et al. 2013).
Conclusion
The obtained information expands our knowledge on the cultivation of the fungus and may further contribute to both mycological and pathological studies involving B. visci isolates and the diseases they cause. Fungus collected from fresh European mistletoe ( V. album) growing on Sorbus aucuparia L. was identified as an anamorph Sphaeropsis visci of the ascomycete Botryosphaeria visci. Our results show that it can be easily introduced into culture and that the growth and morphology of B. visci colonies can vary greatly depending on the media used. The highest mycelium growth and sporulation was found on liquid oatmeal media. The possibility of European mistletoe re-infection in laboratory and field conditions has been evaluated. Our results indicate the biocontrol potential of B. visci against Viscum album infecting various trees.
Acknowledgments
The work was performed under the budget theme of the National Academy of Sciences of Ukraine "Population Biology and Genetics Viscum album L. in Ukraine" (2018-2022), registration number 0118U004067.
The authors are grateful to the Austrian Research and Training Center for Forest, Natural Hazards and Landscape for access to microscope equipment (Dr. Marcela Van Loo).
