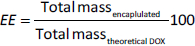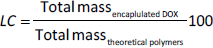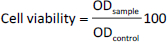Introduction
Doxorubicin (DOX, also known as Adriamycin) has been widely used to treat numerous cancer types since its approval by the FDA in 1974 [1]. The most well-known anticancer mechanism of DOX is the interaction with the topoisomerases II, leading to interference with the DNA replications. Additionally, DOX also induces reactive oxygen species (ROS) via nonenzymatic and enzymatic pathways [2,3]. Consequently, these free radicals cause intracellular oxidative stress, and damage cellular membranes and DNA, although this mechanism is not considered to play a major role in killing cancer cells [4]. Unfortunately, DOX is non-specific distribution due to its low molecular weight drug nature, resulting in myocyte, myelocyte, and hematocyte toxicities [5-7]. DOX-induced damage on intestinal epithelium has also been reported [8]. Besides, the low oral bioavailability of DOX was reported at 1 % due to its absorption through the paracellular pathway [9]. Lowering the cytotoxicity of DOX and enhancement of its oral bioavailability have gained attention to extend the clinical utility of DOX. Recently, scavengers of free radicals showed prevention of DOX-induced cardiotoxicity [10], implying the important role of ROS scavengers in reducing the adverse effects of DOX.
Nanoparticles (NPs) have been widely developed as carriers for drug delivery systems (DDS) to decrease unwanted diffusion, increase drug bioavailability, and control pharmacokinetics [11-13]. Oral administration is one of the most popular pathways to deliver NPs owing to its unique advantages like non-invasion, painlessness, easy administration, and patient compliance. The various NPs, such as liposomes, metal, and polymeric NPs, have been designed for oral drug delivery. However, most techniques still face numerous challenges, such as gastric juice influencing the aggregation state of liposomes [14], decreasing migration, retention in the intestinal mucus layer, and low blood uptake of the therapeutic agents. Moreover, the activation of various enzymes by oral administration also causes instability and leakage of payload, suggesting an efficient reduction of drug carriers and toxicity owing to the exposure of the drug on the gastrointestinal (GI) tract. Therefore, the requirements for oral DDS of cancer treatment are stable dispersion, drug encapsulation in the harsh GI tract, and low toxicity. Recently, polymeric NPs have been applied to improve the oral bioavailability of chemotherapy. For example, DOX-encapsulated enoxaparin sodium-PLGA hybrid NPs significantly increased retention time in the pharmacokinetics study [15]. Moreover, polymeric micelles, a type of polymeric NPs with a core-shell structure, can increase the stability, and permeability in the intestinal epithelium and reduce the degradation of anti-cancer drugs. For instance, DOX-loaded polymeric micelles significantly improved the intestinal absorption rate and systemic circulation time as compared to free DOX [16]. Thus, micelles NPs can be considered potential anti-cancer DDS in oral administration.
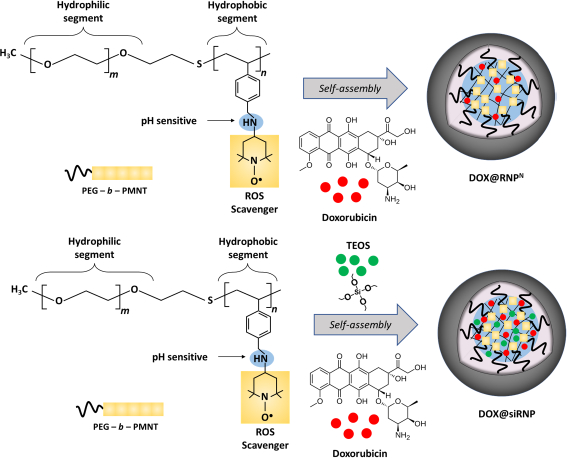
Recently, we have designed two types of core-shell antioxidant NPs, pH non-sensitive (RNP°) and pH-sensitive (RNPN), as ideal oral DDS to solve these problems. RNP° and RNPN were prepared through self-assembly amphiphilic block copolymers containing ROS scavengers (nitroxide radicals, TEMPO) at the side chains of the hydrophobic segment. The ROS-scavenging ability of RNPs is an important factor in decreasing the DOX-induced oxidation stress. Both RNPs indicated highly dispersible and biocompatible properties with a long half-life in circulation compared to free nitroxide radicals. In addition, RNPs have been researched as possible treatments for solid tumours [17,18]. Although RNP° prolonged blood circulation as compared to RNPN [19,20], RNP° was not appropriated in encapsulating drugs inside the core [21]. In contrast, gastric pH is also challenging for RNPN due to its pH sensitivity caused by the protonation of amino groups on the hydrophobic core. Therefore, we have developed silica-installed redox NPs (siRNP) through sol-gel chemistry by the hydrolysis and condensation of tetraethyl orthosilicate (TEOS) with the presence of ammonia during the preparation of RNPN. Crosslinking of silanol groups is expected to improve the stability of NPs and the drug encapsulation efficacy via adsorption on the silica surface and electrostatic interactions between DOX and silica. In this work, we studied the impacts of RNPN and siRNP loading DOX (denoted as DOX@RNPN and DOX@siRNP, respectively) to reduce the side effects of DOX on normal cells and to maintain the cytotoxicity properties of DOX on cancer cells. The obtained result indicated that NPs were nano-size distribution and high dispersion in the physiological conditions. Moreover, the addition of silanol groups significantly improved the encapsulation of DOX and the stability of RNPN in gastric pH. Compared to free DOX treatment, the DOX@RNPN and DOX@siRNP showed lower cytotoxicity against murine fibroblast cells (L929), while they maintained the cytotoxicity on human breast cancer cells (MCF-7) and human hepatocellular carcinoma cells (HepG2).
Experimental
Chemicals
Dimethylformamide (DMF, Sigma-Aldrich, USA), permeable membrane tube (MWCO 3.5 kDa, Spectrum Laboratories Inc., Japan), tetraethyl orthosilicate (TEOS, Sigma-Aldrich, USA), ammonia (NH3, China), Doxorubicin hydrochloride (DOX, Wako, Japan), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT, Roche Diagnostics, Japan), dimethyl sulfoxide (DMSO, China), Dulbecco’s Modified Eagle’s Medium (DMEM, Gibco, USA), fetal bovine serum (FBS, Sigma-Aldrich, USA), and antibiotics (a mixture of penicillin, streptomycin, and neomycin, Sigma-Aldrich, USA) were purchased.
Preparation and characterization of RNPN and siRNP
RNPN was prepared by self-assembly amphiphilic copolymers PEG-b-PMNT, as reported in previous research [18]. Briefly, 15 mg of PEG-b-PMNT were dissolved in 1 mL of DMF and then stirred on a magnetic stirrer. The mixture was then put into a semi-permeable membrane tube before being dialyzed against distilled water for 24 h. To prepare siRNP, a similar method was conducted, except that 50 μL of TEOS and 50 μL of NH3 were added during the stirring process (Figure 1). The average size, polydispersity index (PdI), and size distribution of RNPN and siRNP were then characterized by the dynamic light scattering (DLS, Malvern Zetasizer, UK) system.
The stability of RNPN and siRNP
Phosphate-buffered saline (PBS, 10 mM) was prepared at different pHs, including 3.0, 6.5, and 7.4, to mimic the pH levels of the gastric, tumour extracellular, and physiological environments, respectively. RNPN and siRNP were then diluted in the prepared buffers. The stability of RNPN and siRNP was evaluated by measuring the change in light scattering intensity using the DLS system at 25 °C for 2 h.
Preparation and characterization of DOX@RNPN and DOX@siRNP
DOX@RNPN and DOX@siRNP were prepared by co-dissolving 1 mg of DOX and 15 mg of PEG-b-PMNT in 1 mL of DMF. Similar steps were followed in the preparation of RNPN and siRNP, respectively (Figure 1). Additionally, various ratios of TEOS:NH3 (200 μL TEOS: 200 μL NH3, 100 μL TEOS: 100 μL NH3, 50 μL TEOS: 50 μL NH3, 100 μL TEOS: 50 μL NH3, 200 μL TEOS: 50 μL NH3) were examined to optimize the size of DOX@siRNP. The DLS system was used to characterize the average size, PdI, and size distribution of DOX@RNPN and DOX@siRNP.
Drug encapsulated efficiency and loading capacity of DOX@RNPN and DOX@siRNP
The encapsulation efficiency (EE / %) and loading capacity (LC / %) of DOX@RNPN and DOX@siRNP were investigated based on the amount of loaded DOX evaluated by a microplate reader (Thermo Fisher Scientific, USA) with a calibration curve of DOX measured absorbance at 480 nm. The EE and LC were calculated by usingequation (1) and(2):
Release profile of DOX@RNPN and DOX@siRNP
2 mL of DOX@RNPN or DOX@siRNP were loaded into separate dialysis bags and placed in different beakers under stirring conditions, each with distinct pH including 3, 6.5, and 7.4. At predetermined time points (0, 1, 3, 6, 9, 20, and 24 h), samples were collected and the absorbance at 480 nm was measured to evaluate the amount of drug released from the NPs.
Cytotoxic assay
The murine fibroblast cells (L929), human breast cancer cells (MCF-7), and human hepatocellular carcinoma cells (HepG2) were obtained from the American Type Culture Collection (ATCC, USA). The cells were cultured in DMEM containing 10 % FBS and 1 % antibiotics (a mixture of penicillin, streptomycin, and neomycin) at 37 °C and 5 % CO2. The cells were then seeded on 96-well plates (104 cells/well) and incubated for 24 h at 37 °C and 5 % CO2. The tested samples, including RNPN, siRNP, DOX, DOX@RNPN, and DOX@siRNP, were added to each well to obtain final concentrations of DOX at 0.25, 0.5, 1, 2.5, and 5 μg/mL, and incubated for 24 h. After that, the medium was removed, and MTT solution was added to each well and continuously incubated for 4 h. Finally, DMSO was added to dissolve crystals of formazan inside the cells, and the plates were measured absorbance at 540 nm by a microplate reader (Thermo Fisher Scientific, USA). The cell viability (%) was calculated by usingequation (3):
where ODsample is the absorbance of the sample and ODcontrol is the absorbance of the solvent.
Results and discussion
Characterization of RNPN and siRNP
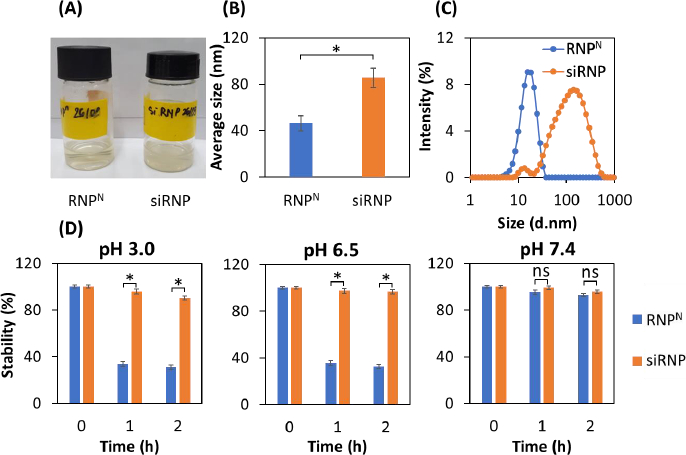
RNPN and siRNP were prepared by self-assembly amphiphilic copolymers PEG-b-PMNT in DMF, followed by dialysis against distilled water for 24 h to remove the organic solvent. The results showed that both RNPN and siRNP were transparent (Figure 2A), with average sizes of 45.5 and 85.5 nm, respectively (Figure 2B). Furthermore, the PdI of both NPs was less than 0.3, indicating a narrow size distribution. The increase in the size of siRNP as compared to RNPN might be explained by a sol-gel reaction of TEOS in the RNPN core [22]. The size of siRNP was approximately 85 nm after the condensation reaction, suggesting no significant aggregation (Figure 2C), even though silica particles formed by sol-gel can reach up to 2000 nm. We next examined the influence of different pH buffers on the stability of NPs [23]. As shown inFigure 2D, RNPN disintegrated under acidic pH (3.0 and 6.5) and stabilized at pH 7.4. In contrast, siRNP was stable regardless of pH change. This might be explained that the amino groups of the PMNT segment disintegrated due to their protonation at low pH. However, adding silica would maintain the structure of NPs at acidic pH, resulting in enhanced stability.
Characterization of DOX@RNPN and DOX@siRNP
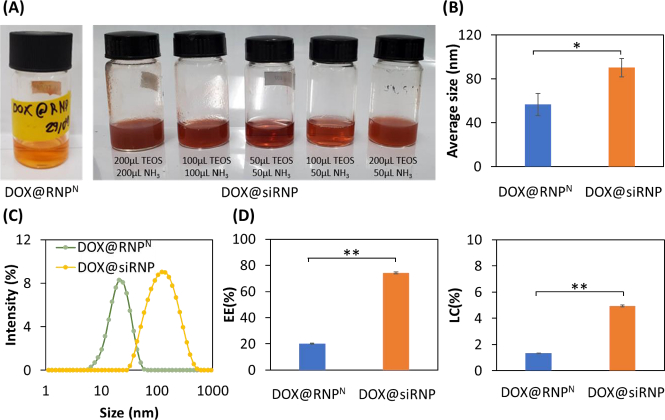
DOX@RNPN and DOX@siRNP were similarly prepared as RNPN and siRNP, respectively, except for the addition of DOX during the preparation process. Different concentrations of NH3 might result in a change in the size of silica NPs despite its catalyzation role in sol-gel reactions [24]. Thus, different ratios of TEOS:NH3 were examined to prepare DOX@siRNP with the nano-size distribution. As shown inFigure 3A, DOX@RNPN, DOX@siRNP with ratios of 50 μL TEOS: 50 μL NH3 and 100 μL TEOS: 50 μL NH3 were transparent, resulting in the nano-size distribution as compared to other samples with turbidity in solution and micro-size distribution (data not shown). Hence, DOX@siRNP with a ratio of 50 μL TEOS: 50 μL NH3 was chosen for further experiments because of its suitable size. As compared to NPs, the size of both DOX@RNPN and DOX@siRNP significantly increased to 56 and 90 nm, respectively, implying the encapsulation of DOX in NPs (Figure 3B). The critical cut-off point for endocytosis has been estimated from 20 to 500 nm for M cells and from approximately 50 to around 100 nm for enterocyte cells [25]. Therefore, DOX@RNPN and DOX@siRNP showed the potential to increase the oral bioavailability of DOX. Moreover, the PdI of both NPs was around 0.3, indicating the narrow size distribution even after drug encapsulation (Figure 3C). Conventionally, polymeric NPs exhibit a low EE by physical entrapment of the drug (less than 10 %) [26-28]. For example, the LC of DOX-loaded silica NPs was reported at 4.8 % [29]. As shown inFigure 3D, the EE was 20.3 and 74.9 %, and the LC was 1.5 and 5 % for DOX@RNPN and DOX@siRNP, respectively. This result indicates the appropriate design of the DOX encapsulation because the silanol groups in the core of siRNP significantly improved the EE and LC of DOX. This result could be explained by the pKa of the amino group in DOX being around 9.9, thus most DOX molecules are in the positively charged form at physiological pH [30]. Consequently, silanol moieties possessing a negatively charged surface could interact with DOX by electrostatics, suggesting the enhancement in EE and LC of DOX@siRNP.
Release profile of DOX@RNPN and DOX@siRNP
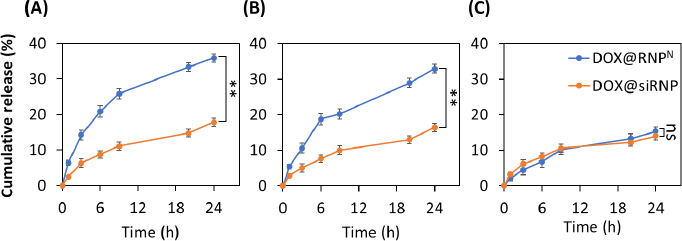
The release kinetics of DOX@RNPN and DOX@siRNP were evaluated under different pH buffers, including pH 3.0, 6.5, and 7.4, which mimic the gastric, tumour extracellular, and physiological pH, respectively. Gastric pH is one of the challenging barriers for oral drug delivery systems. As shown inFigure 4A, DOX@siRNP was released significantly slower than DOX@RNPN (17.8 % compared to 35.9 %) after 24 h, even though both experienced protonations of amino groups at the hydrophobic core. However, more than 80 % of DOX@RNPN and 90 % of DOX@siRNP were retained after 6 h, suggesting their potential for in vivo application as they can withstand the maximal transit time of the stomach in the human body. The extracellular pH of tumors, which commonly drops around 6.5-7.0, is mainly caused by anaerobic glycolysis in hypoxia [31]. As shown inFigure 4B, the release profiles of DOX@siRNP and DOX@RNPN at pH 6.5 were 16.4 and 33.0 %, respectively. These results are consistent with the previous stability study. It is evident that DOX@siRNP was less impacted by acidic pH than DOX@RNPN due to the ability of nano-silica to stabilize the structure. After 24 h at physiological pH, DOX@RNPN and DOX@siRNP leaked at 14 and 15 %, respectively (Figure 4C). Similarly, several studies have reported that the release profile of micelle NPs with pH-sensitive groups is higher at acidic pH than at neutral pH [32,33].
In vitro cytotoxicity
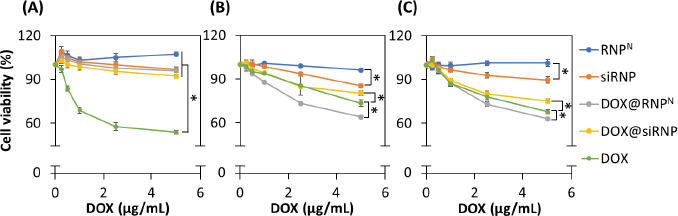
The cytotoxicity of NPs was assessed through MTT assay on normal murine fibroblast cells (L929) and cancer cells, including human breast cancer cells (MCF-7) and human hepatocellular carcinoma cells (HepG2). In addition to the well-known anticancer mechanism, which involves interaction with the topoisomerases II to interfere with DNA replication, it also induces the over-expression of free radicals to kill cells. Therefore, RNPN with TEMPOL, a ROS scavenger in the hydrophobic core, was used to encapsulate DOX to reduce its side effects in an in vitro model. As shown inFigure 5A, DOX indicated dose-dependence, with approximately 50 % of L929 cell death at a concentration of 5 μg/mL DOX, while the NPs significantly enhanced cell viability, almost equal to the control sample. Despite dose-dependent toxicity observed in DOX@RNPN and DOX@siRNP, there was more than 90 % of cell viability after 24 h of incubation due to the scavenging ability of TEMPO groups. In addition, silica NPs have been reported to cause cytotoxicity due to the overproduction of ROS [34], leading to lower cell viability of siRNP compared to RNPN. However, siRNP treatment exhibited more than 90 % of cell viability, suggesting the biosafe profile of NPs. Similarly, anticancer effects of DOX were observed on human breast cancer cells (MCF-7) and human hepatocellular carcinoma cells (HepG2), with dose dependence (Figure 5B andFigure 5C). Remarkably, DOX@siRNP caused fewer cell deaths (around 20 and 25 %) compared to (27 and 32 %) at 5 μg/mL of free DOX treatment against MCF-7 and HepG2, respectively. This result may be due to DOX being kept inside the core of siRNP, resulting in less exposure to cells, although encapsulation of DOX in NPs was expected to be more effective at entering cells. In contrast, the anticancer effect caused by DOX@RNPN (5 μg/mL of DOX) was 36 and 37 % for MCF-7 and HepG2, respectively. It is obvious that the cumulative release of DOX@RNPN was faster as compared to DOX@siRNP, resulting in higher cytotoxicity when entering cells.
Conclusions
In this study, non-silica and silica redox NPs (RNPN and siRNP, respectively) with ROS scavenging moieties on hydrophobic side chains were prepared to encapsulate DOX to reduce its adverse effects. The DOX@RNPN and DOX@siRNP were successfully prepared with several tens nm distributions and stable dispersion. The obtained results indicated that the addition of silanol groups significantly improved the stability of RNPN in acidic pH and the encapsulation efficacy of DOX into siRNP. Due to its pH-sensitive character, DOX@RNPN exhibited a faster release of DOX under acidic pH environments as compared to DOX@siRNP. Both DOX@RNPN and DOX@siRNP suppressed the cytotoxicity of DOX against normal fibroblast cells while they exhibited the anticancer activity to induce cell death in liver cancer and breast cancer cell lines. Therefore, DOX@RNPN and DOX@siRNP could be considered potential nanomedicines for cancer treatments.